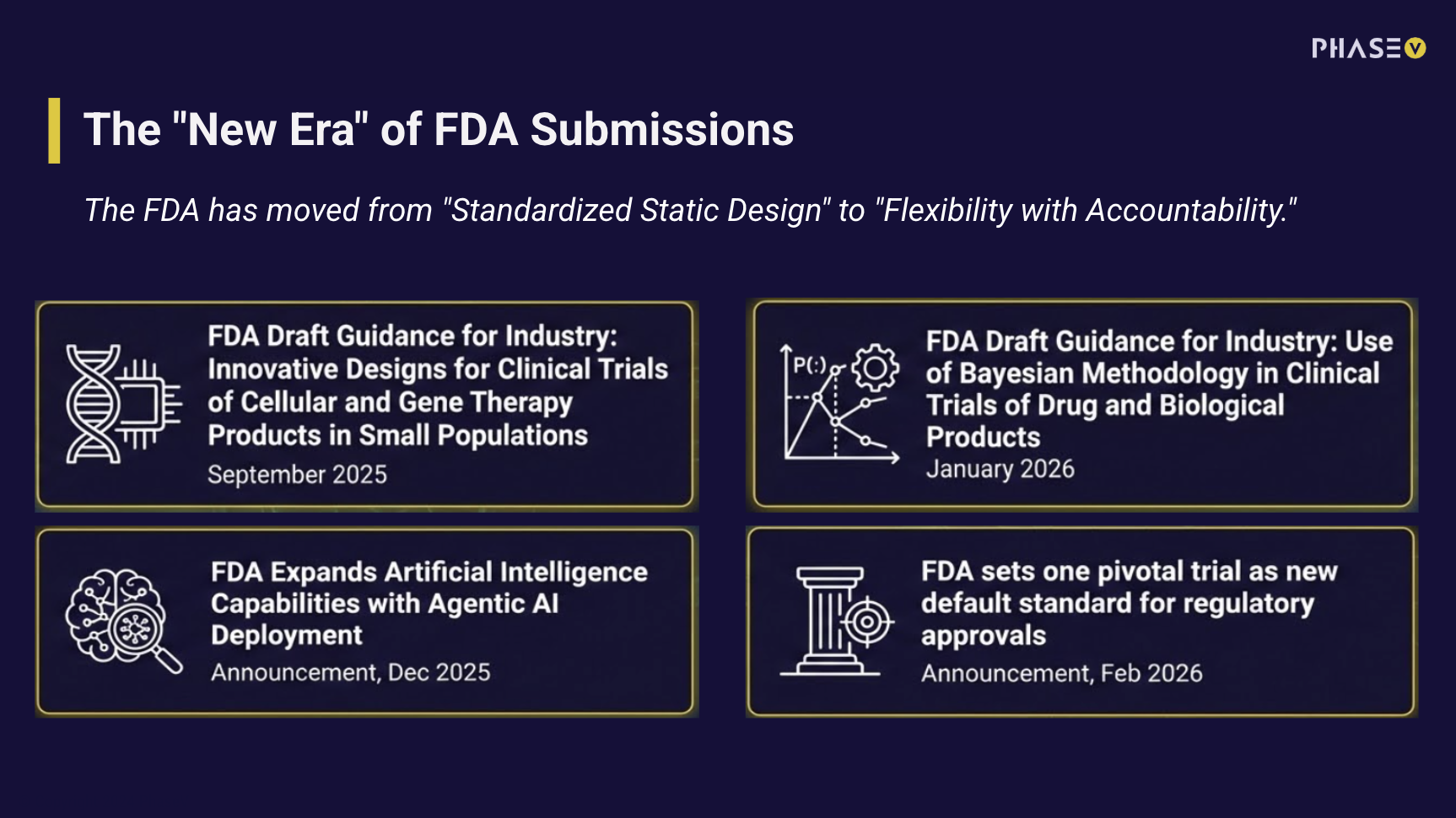
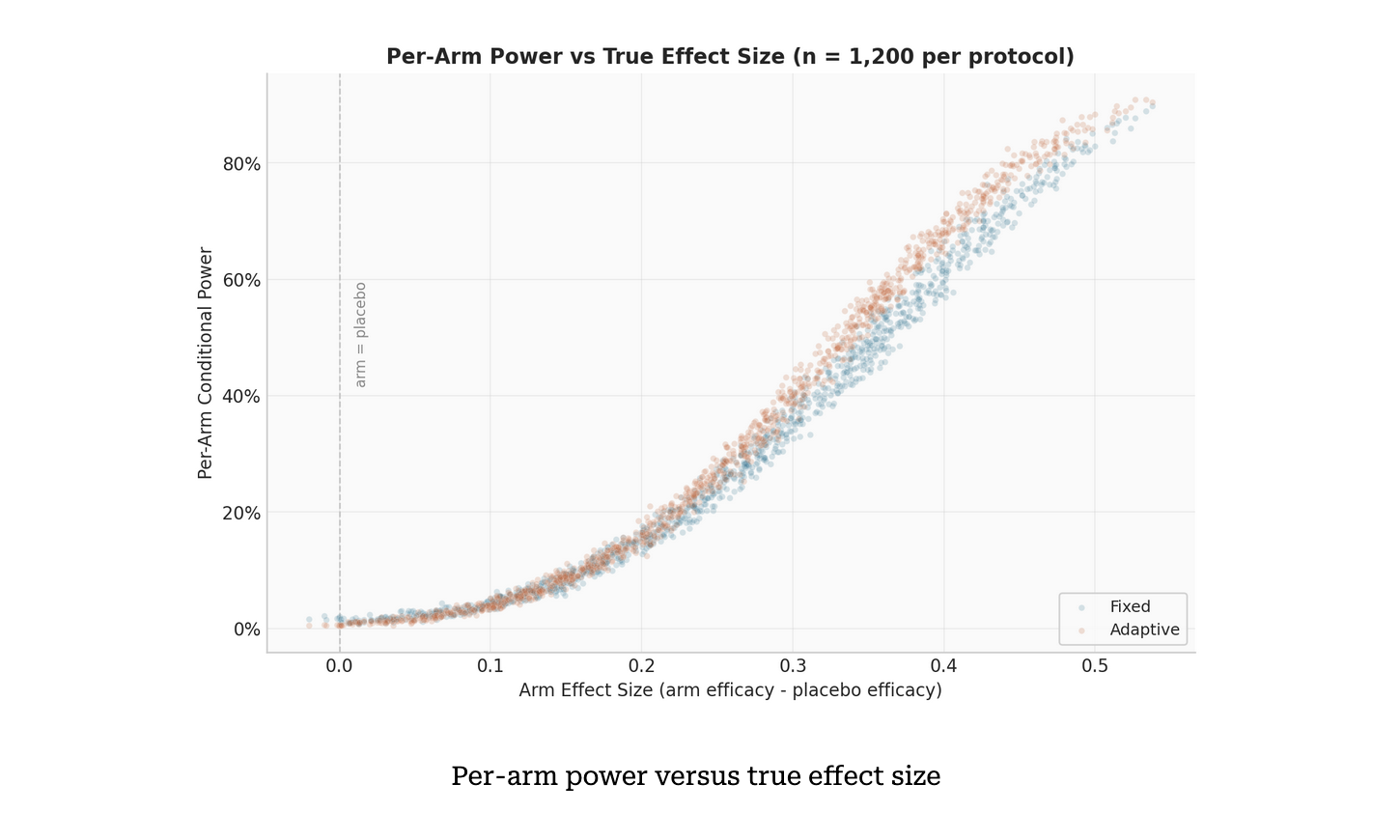
You Can't Model What You Can't See: Clinical Trial Transparency and the PoS Problem
The FDA’s recent push to nudge sponsors into reporting missing clinical trial results highlights a long-standing issue in drug development: we still operate with a systematically incomplete picture of failure.
Roughly 30% of trials that likely fall under mandatory reporting requirements have not submitted results. In a fiery statement, FDA Commissioner Marty Makary, M.D., M.P.H. makes a bold claim:
"Far too often, companies are suppressing unfavorable clinical trial results and keeping them secret from patients and the scientific community. Those sponsoring clinical trials have an ethical obligation to make results public regardless of the data’s influence on the company’s share price"
That’s not just a transparency problem—it’s a modeling problem. When failed or inconclusive trials go unreported, the industry loses the ability to rigorously decompose why programs fail. Was it a lack of efficacy? Safety signals? Trial design? Patient selection? Endpoint choice? Without that granularity, failures collapse into a single opaque category: “didn’t work.”
This missing data directly undermines our ability to estimate probability of technical success (PTS) and probability of regulatory success (PRS). These probabilities are not abstract—they are built on historical analogs. But if the historical record is biased toward successful or selectively reported outcomes, then any downstream estimation is also biased. We end up overestimating success rates, underestimating risk, and misallocating capital.
At PhaseV we are building sophisticated statistical imputation methods, combined with LLM based data curation to overcome this problem - spending time, resources and effort to overcome a problem that could have been avoided.
More importantly, the absence of detailed negative data prevents true failure mode decomposition. In a well-instrumented system, we should be able to say, for example, that a given mechanism fails 40% of the time due to target biology, 30% due to trial design, and 30% due to execution. Instead, we’re left with coarse heuristics and noisy proxies. This makes it difficult to learn systematically across programs, indications, and modalities.
The consequences ripple outward. Investors price assets with incomplete risk profiles. Clinical teams design trials without full visibility into prior pitfalls. Regulators and researchers lack the ability to contextualize new evidence against a comprehensive baseline. Ultimately, patients bear the cost through slower, less efficient development.
The FDA’s reminder letters are a step, but as critics note, they may be more symbolic than substantive without enforcement. The real opportunity is not just compliance for compliance’s sake, but the creation of a complete, high-resolution dataset of both success and failure.
Drug development doesn’t just need more data, it needs the right data. And until failed trials are consistently reported with sufficient detail, our understanding of risk will remain fundamentally constrained.